Focused on rehabilitation equipment, covering furniture, motion control, and functional evaluation series solutions
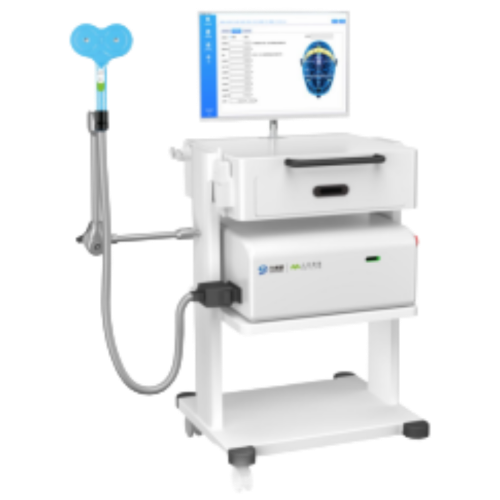
Magnetic stimulation device KNZ-TMS-C100
Model:KNZ-TMS-C10
Medical device registration certificate number: Hubei Medical Device Registration No. 20252095779
 Sales Tel:027-59702088
Sales Tel:027-59702088
 Product Details
Product Details
Product Details
Non-invasive target stimulation, efficient neural regulation
√ 100KHz sampling rate dual-channel MEP module, 3 waveform processing methods.
√ Equipped with multi-mode TMS management software, suitable for clinical use of transcranial magnetic and pelvic magnetic stimulation.
√ Built-in treatment prescription and brain positioning map, allowing parameter customization and automatic report generation.
√ Features dual cooling systems of liquid cooling and air cooling, with real-time flow rate and temperature monitoring.
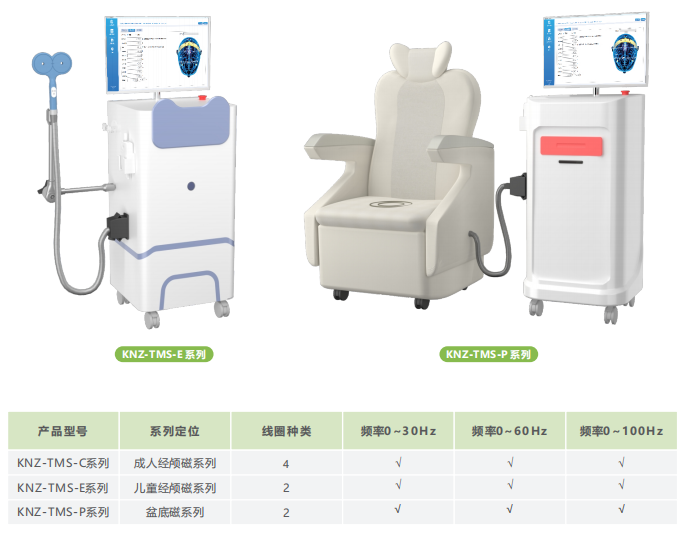
Applicable departments: Rehabilitation Medicine Department, Neurology Department, Geriatrics Department, Obstetrics and Gynecology Department, Pediatric Rehabilitation Department, Psychiatry Department, etc.









